|
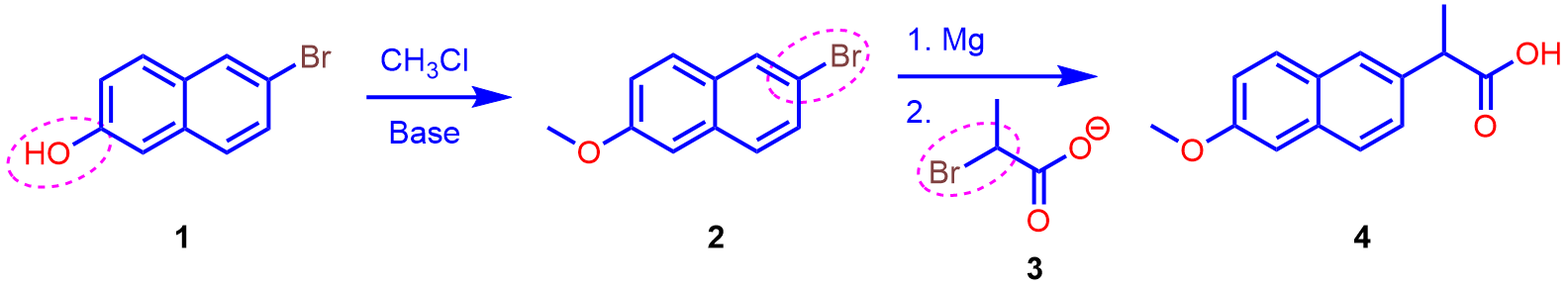
Identify the reaction that is occurring.
(Check for chemoselectivity.)
|
The first, and perhaps the most difficult, step is to figure out what transformation is occurring.
Refer to your learning resources to learn about the reagent(s) in the reaction scheme, and the reactions that the functional groups in the starting materials undergo.
You should be able to identify a unique transformation that occurs when one or two of the functional groups react with the reagents (if any) under the conditions shown in the scheme.
An example problem is shown below.
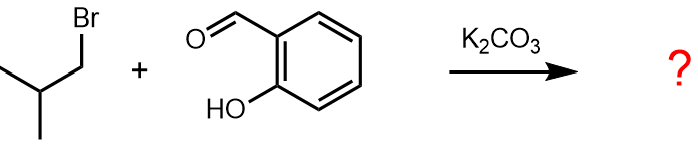
Review all the functional groups in the starting materials and assess if they are likely to react under the conditions.
One of the starting materials in the problem is an alkyl bromide and one of the most common reactions of such compounds is that the bromide undergoes substitution by nucleophiles.
The other starting material is a phenol, which is moderately acidic, and the reagent is a base (potassium carbonate) which would deprotonate the phenol to give a phenolate anion, which could then act as a nucleophile.
There is also an aldehyde functional group in the starting materials, but a review of the reactions of aldehydes will show that this is unlikely to react under these conditions, i.e. this is a chemoselective transformation.
Thus, the likely reaction is nucleophilic substitution to give an ether, i.e. R¹–Br + HOR² → R¹OR².
Note the overall bonding changes that occur in the transformation.
In this example, (i) the bond from the nucleophilic atom to H is broken, (ii) the nucleophilic atom becomes bonded to the carbon bearing the bromine, and (iii) the carbon to bromine bond is broken.
Other functional groups may be present, but not affected, in which case the reaction is said to be chemoselective.
In this example, the aldehyde is inert under the conditions of the reaction.
'Inert' functional groups are indicated with dashed gray ovals in the schemes.
|
|
Identify the reaction site(s) in the starting material(s).
(Check for regioselectivity.)
|
Having deduced what the transformation is, identify the reaction site(s), the atom(s) at which the bonding changes will occur and that will be retained in the product.
The other atoms in the starting material(s) will not undergo any changes in the reaction.
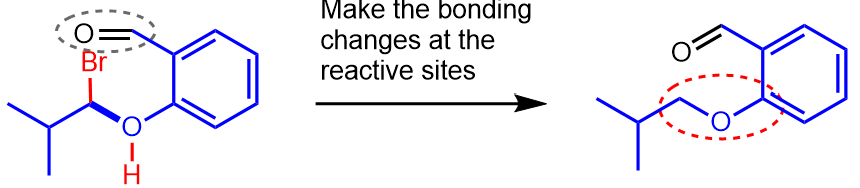
In our example, the reaction sites are the oxygen of the phenol and the carbon which undergoes substitution with displacement of the bromide (bright blue circles below).
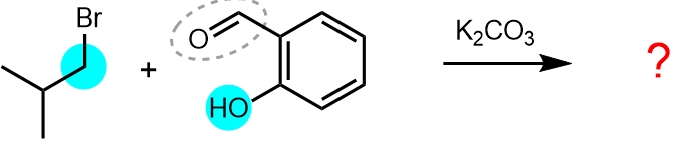
Make sure that you identify all instances of the reactive functional groups and reactive sites, e.g. reduction of a diketone will involve two reaction sites.
For most reactions, identifying the reaction sites is quite easy once the nature of the transformation has been established.
However, for some important reactions the reacting functional group can react at more than one site, so the new functional group, or new substituent, can be attached at more than one position.
In such reactions regioselectivity is an issue.
Some common reactions that often give regioselectivity issues are additions to alkenes, electrophilic aromatic substitutions, and alpha-substitution of ketones.
For example, the aromatic compound on the left below undergoes regioselective electrophilic aromatic substitution at the atom highlighted in bright blue, with minor amounts of products formed by reaction at the atom in pale blue.
The ketone in the centre has two different alpha positions, and reactions at the atom highlighted in bright blue generally predominate.
Finally, the alkene on the right is unsymmetrical and undergoes addition of unsymmetrical reagents with Markownikov, or anti-Markownikov, regioselectivity depending on the reagent.
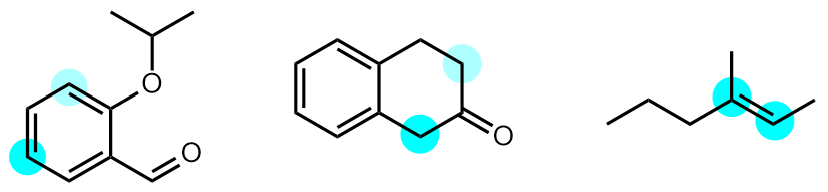
|
|
Draw the skeleton of the product and add functional groups that remain unchanged in the transformation.
|
The 'skeleton' of the product is the framework of carbons atoms, and other 'heavy' (non-hydrogen, most commonly O or N) atoms linking groups of carbon atoms.
The distinction between the 'functional groups' and the 'skeleton' is somewhat arbitrary; what is important is that you correctly draw the skeleton and all functional groups when drawing a product.
Take care to draw the correct number and arrangement of C, and linking O and N, atoms so that the product structure is correct.
In order to focus on getting the skeleton right, leave the other bonds at the reaction sites unchanged at this stage, the necessary changes will be made in the next step.
If the transformation is a 'functional group interconversion' that does not involve addition of carbon atoms from another starting material, the skeleton remains unchanged from the starting material and is relatively easy to draw.
If the transformation involves joining of the skeletons of two starting materials together (formation of a new C–C, C–O, C–N or C–S bond), drawing the skeleton of the product is non-trivial.
One way to do this is to carefully copy the skeletons of the two starting materials, and draw lines between reaction sites that are becoming bonded to each other, to form the skeleton of the product.
If the transformation involves a rearrangement, the skeleton will, by definition, be changed, and you will need to understand the mechanism of the reaction to predict the skeleton of the product.
The example below shows a Beckmann rearrangement involving skeletal reorganisation from a six- to a seven-membered ring.
The skeletal rearrangement is indicated by dashed red (broken) and blue (formed) bonds in the starting material.
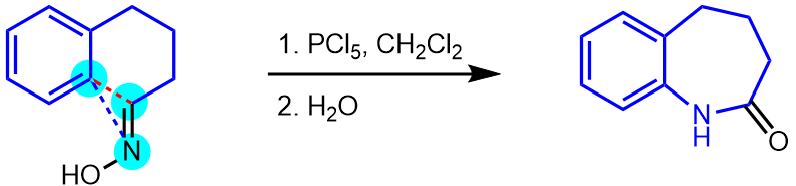
Then add all the functional groups that remain unchanged in the transformation, making sure that they are in the same positions as in the starting materials.
That includes 'inert' functional groups that are not involved at all, but also groups that are involved, but do not change, because the reaction takes place at an adjacent carbon.
A very common example is reaction at carbons alpha to carbonyl groups, in which a substituent is introduced at the alpha position but the carbonyl group remains intact.
In our example, the skeletons of the two starting materials are joined via a new C–O bond, and the aldehyde group is added, as shown below (remember that we have not yet made the bonding changes at the reactive sites).
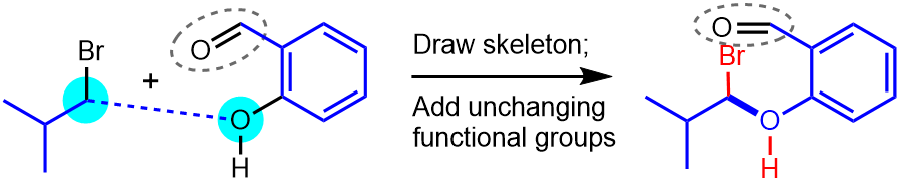
|
|
Make the required changes to the bonding at the reaction sites to the skeleton, so that the functional groups that are formed in the reaction are correct.
|
Refer to the bonding changes that take place at each reaction site in the transformation, and make the appropriate bonding change(s) at the reaction site(s) in the specific example given in the problem.
You should now have the functional groups that are formed in the transformation to the products.
In our example the O of the phenol will lose a H, the C–Br bond will be broken, giving the ether (dashed red circle) shown below as the product of the reaction.
|
|
Draw the stereochemistry at chirality centres and/or alkenes.
(Check for stereoselectivity.)
|
First, remember that a stereogenic centre is an 'atom within a molecule bearing groups such that interchanging any two of them leads to a stereoisomer of the original molecule'.
There are two common types of stereogenic centres in organic compounds:
(i) chirality centres, tetrahedral atoms with four different groups attached which give rise to two possible configurations (R & S), and
(ii) the carbons of alkenes that exhibit cis/trans isomerism.
In order to correctly draw the stereochemistry of product molecules that contain stereogenic centres, you will need to consider three situations that may arise:
● Stereogenic centres that have remained unchanged in the transformation.
These are quite easy to deal with; take care to draw the same configurations of 'inert' chirality centres and stereogenic alkenes in the product(s) as in the starting material(s).
● Chirality centres formed by bonding changes at chirality centres in the starting materials.
There are three possible outcomes, stereoselective inversion of configuration, stereoselective retention of configuration, and formation of mixtures (low/no stereoselectivity).
Refer to learning materials to find out which outcome is expected for the type of reaction shown in the problem.
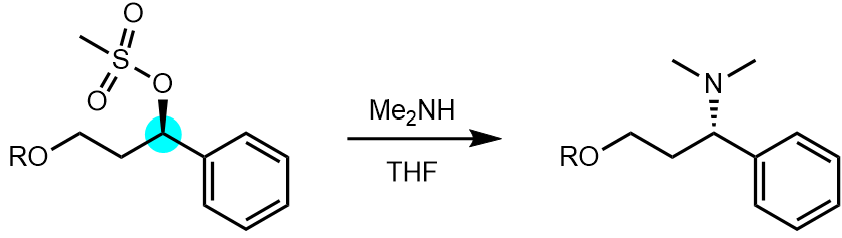
For example, reaction of the mesylate below with an amine gave substitution with complete inversion of configuration (indicative of the SN2 mechanism).
● New chirality centres formed by bonding changes at atoms that were not chirality centres.
At the moment the problems in ChemInteractive do not involve selectivity for one configuration at new chirality centres.
However, when two new chirality centres are formed their relative configurations are checked.
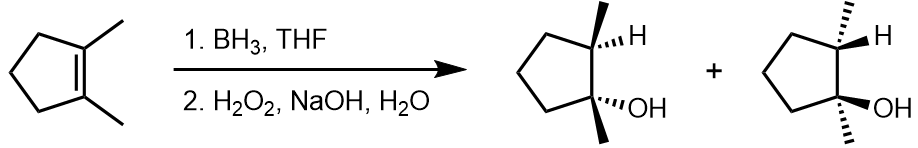
This often arises in additions to alkenes, in which the relative configurations of the two new centres depends on whether the addition was syn or anti.
For example, hydroboration/oxidation of 1,2-dimethylcyclopentene gave syn addition exclusively, though the product was, of course, a racemic mixture.
|